Why Xypex for Marine Structures

Meeting the Challenge
Protecting Marine Structures
Reinforced concrete marine structures are constantly under attack by the destructive effects of moisture and chloride-induced corrosion. Without proper protection, the structural integrity of a marine structure is soon compromised, leading to expensive remediation efforts and a shortened life span. Once the moisture and chlorides have reached the reinforcing steel, an expansive oxidation process begins to take place causing the formation of cracks and spalling in the concrete. When cracking takes place and is combined with weathering effects such as freeze/thaw damage or accelerated corrosion in hot weather climates, this deterioration takes place at a faster pace.
With over 50 years of experience in 90 countries around the world, Xypex Crystalline Technology has been used in marine structures to waterproof, protect, repair and enhance the durability of concrete subject to water and chloride attack. In this challenging environment, where structures are also exposed to sulphate attack, carbonation, alkali-aggregate reaction, abrasion and freeze thaw cycles, Xypex is a highly respected partner in extending the service life of marine construction.
Xypex products play a key role in the waterproofing and protection of concrete against water penetration, chloride ion attack, cracking, carbonation, sulphate attack, Alkali Aggregate Reaction and freeze/thaw damage – problems typically associated with the reduced service life of marine structures.

Water Permeability & Corrosion
The primary purpose of waterproofing concrete marine structures is the protection of reinforcing steel from the damaging effects of corrosion. The nature of concrete and the problems associated with placement and consolidation means having to deal with permeability issues permitting the penetration of water into the substrate and through to the reinforcing steel. With the presence of oxygen this can initiate corrosion.
This permeability facilitates the entry and diffusion of chlorides into contact with the reinforcing steel. The resulting loss of alkalinity and hence the passivating layer allow for an electrochemical process culminating in corrosion of the reinforcing steel and the expansive disruption of the concrete substrate.

Cracking
Cracks in the concrete are the most obvious means by which water and damaging chemicals can enter a structure. These cracks are formed in a number of different ways but the most common are drying shrinkage, thermal cracking, strain formed cracks, settlement cracks and plastic shrinkage cracking in the slab.

Surface Deteroriation
The principal cause of surface degeneration is the impact of silt, sand, gravel and other solids impacting and rolling against the concrete surface. This causes deterioration in the long term, resulting in some form of surface rehabilitation during the service life of the structure.

Carbonation
Carbon dioxide in the air reacts with calcium hydroxide in the concrete to form calcium carbonate which reduces the alkalinity of the concrete. Below a pH of 10, the rebar’s thin layer of surface passivation dissolves and corrosion of the reinforcing steel takes place at an accelerated rate.

Sulphate Attack & Alkali Aggregate Reaction
Where sulphates are present in water or soils, the permeability of concrete and the presence of water allows sulphate ions to diffuse into the concrete and create an expansive reaction causing spalling and deterioration.
A similar effect is caused by Alkali Aggregate Reaction whereby the presence of water in concrete permits a reaction between silica in certain aggregates and the alkalis in cement.
Xypex Crystalline Technology
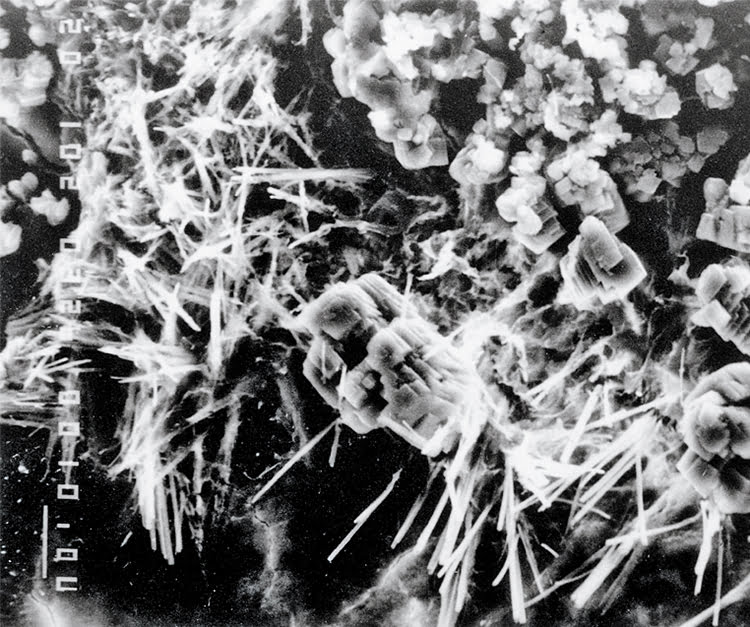
Xypex products use the natural porosity of concrete and chemical diffusion to penetrate its pores and capillaries. Inside the concrete, Xypex chemicals react with un-hydrated cement particles and the by-products of cement hydration to form a non-soluble crystalline structure deep within the substrate. In this condition, the concrete becomes impermeable, preventing the penetration of liquids and chemicals from any direction even under extreme hydrostatic pressure. The chemical resistant properties of the crystalline structure will mitigate the attack of chlorides and sulfates. In prolonging the durability of concrete, Xypex has also proven to be effective against the effects of carbonation and Alkali Aggregate Reaction as well as having the ability to self-heal static cracks up to 0.5 mm (0.02 in). Xypex also improves the freeze-thaw durability of concrete.
The Xypex Advantage
Xypex Crystalline Technology works inside the concrete, thereby avoiding the problems typical of traditional barrier products.
- Permanent and reactivates whenever water is present
- Protects against chlorides
- Self-heals static cracks up to 0.5 mm (0.02 in)
- Protects against sulphate attack and Alkali Aggregate Reaction (AAR)
- Reduces the rate of carbonation
- Not subject to deterioration problems encountered by surface coatings and membranes
Proven Performance Worldwide
Comprehensive quality systems and standards along with thorough testing in the lab and the field have resulted in Xypex’s highly respected position in the concrete industry. Xypex has been extensively tested by independent testing laboratories in the U.S., Canada, Australia, Japan, Europe and other countries.





Xypex Admix for New Concrete Construction
Xypex Admix is the preferred choice for installing Xypex Crystalline Technology into most new concrete marine structures. Because Xypex Admix is blended into the mix at the time of batching, it becomes an integral part of the entire concrete matrix, thus reducing the potentially damaging effects of water penetration, chloride and sulfate attack, carbonation and Alkali Aggregate Reaction. The addition of Xypex Admix to concrete is a highly effective method of enhancing the durability of concrete structures.
Xypex Admix Advantages
- Permanent integral waterproofing
- Enhances concrete durability
- Value engineering
- Non-toxic
- Non-combustible
- Resists damaging effects of water penetration and chemical attack

Discover the full potential
of Additive products.



Rehabilitation & Repair
Xypex’s coating systems and repair products enable owners, engineers and contractors to economically and confidently repair structures that have been damaged due to the effects of chloride attack, carbonation, Alkali Aggregate Reaction or surface deterioration through abrasion and freeze/thaw. Xypex Concentrate and Modified are applied as slurry coatings to the surface of the concrete. Unlike other materials that need a dry substrate, Xypex products require a moist surface – a condition typical of marine structures. This type of environment is conducive to the Xypex Crystalline process. Xypex Patch’n Plug, Concentrate Dry-Pac, and Megamix products are specifically designed to permanently repair concrete defects such as static cracks and faulty cold or construction joints. Xypex Megamix returns structural integrity to severely damaged concrete, whilst maintaining the same protective properties of Xypex-treated concrete.
Xypex Coating Advantages
- Doesn’t require a dry surface
- Apply to either side of the concrete
- Won’t puncture, blister or tear
- No costly surface priming or leveling
- Sealing, lapping or finishing is not required
- No need for protection during backfilling
- Permanent waterproofing
- Enhanced concrete durability
- Doesn’t contain VOCs
- Non-toxic and non-combustible
- Can be applied safely in confined spaces
Other Accessory Products are also available.
SEE XYPEX PRODUCTS

Find the right product
for your project.




Singapore Marina Barrage

Sandy Bay Seawall

Conrad Maldives Resort & Spa

Jacksonville Electric Authority Wharf

Lascelles Wharf

Flinders Ports Berth 8 Outer Harbour Grain Wharf

Port of Guayaquil

Australian Marine Complex

Mayport Naval Station